Ethical Principles and Publication Policy
Publication Ethics Principles
Publication Ethics can be defined as a self-regulatory mechanism that insists on integrity on behalf of authors, reviewers and publishers to establish higher standards of editorial processing. Ethical standards for publication exist to ensure high-quality scientific publications, public confidence in scientific findings, and respect for people's opinions.
- Honest researchers do not plagiarise.
- They do not misattribute sources.
- They do not hide objections they cannot refute.
- They do not distort opposing views.
- They do not destroy or conceal data.
Peer-reviewed studies are studies that support and realise the scientific method. At this point, it is of great importance that all parties involved in the publication process (authors, readers and researchers, publisher, referees and editors) comply with ethical principles. RumeliDE journal adheres to national and international standards on research and publication ethics. It complies with the Press Law, the Law on Intellectual and Artistic Works and the Directive on Scientific Research and Publication Ethics of Higher Education Institutions. RumeliDE has adopted the International Ethical Publishing Principles published by the Committee on Publication Ethics (COPE), Directory of Open Access Journals (DOAJ), Open Access Scholarly Publishers Association (OASPA) and World Association of Medical Editors (WAME). It also undertakes to comply with the decisions of the Turkish Editors' Workshop.
- Press Law (National Legislation)
- Law on Intellectual and Artistic Works (National Legislation)
- Higher Education Institutions Scientific Research and Publication Ethics Directive (National Legislation)
- Transparency and Best Practice Principles in Academic Publishing (International Criteria)
- Turkey Editors' Workshop Decisions (National Criteria)
Replay
Republishing is the publication of the same article or substantially similar articles in more than one journal. The editor sends such an article back without reviewing it. Thereafter, the editor may impose an embargo for a certain period of time on the author who has attempted to republish, publicise this situation in the journal in which the author has previously published (perhaps by simultaneous announcement with the editor of the journal that published the previous article), or apply all of these measures together.
Simultaneous submission of the same work to more than one journal
Authors may not submit the same article to more than one journal at the same time. If the editor learns of possible simultaneous submission, he/she reserves the right to consult with the other editor(s) receiving the manuscript. In addition, the editor may return the manuscript without review or reject the manuscript without considering the reviews, or take this decision in discussion with the other editor(s) involved, and may decide not to accept submissions from authors for a certain period of time. It may also write to the authors' employers or take all of these measures together.
Control to Prevent Plagiarism
Plagiarism is presenting the ideas, methods, data, applications, writings, figures or works of others as one's own work in whole or in part without citing the authors in accordance with scientific rules.
RumeliDE scans all submitted articles to prevent plagiarism. The studies submitted for review are checked for plagiarism using Turnitin & Ithenticate software. The similarity rate is expected to be less than 25%. The main measure of similarity is the author's compliance with the rules of citation and citation. If the similarity rate is 1%, but citation and quotation are not done properly, plagiarism may still be in question. In this respect, citation and quotation rules must be known and carefully applied by the author.
Plagiarism, duplication, false authorship/ denied authorship, research/data fabrication, article slicing, sliced publication, copyright infringement and concealment of conflict of interest are considered unethical behaviours. All articles that do not comply with accepted ethical standards are removed from publication. This includes articles containing possible irregularities and non-conformities detected after publication.
Forgery
To produce data that is not based on research, to edit or change the work presented or published on the basis of unreal data, to report or publish them, to show a research that has not been done as if it has been done.
Falsifying research records and data obtained, showing methods, devices and materials that were not used in the research as if they were used, not evaluating data that do not comply with the research hypothesis, manipulating data and/or results to fit the relevant theory or assumptions, falsifying or shaping the research results in line with the interests of the persons and organisations supported.
Protection of Personal Data of Participants
RumeliDE requires that all research involving personal or sensitive data or materials relating to human participants that are not legally publicly available be subject to formal ethical review.
Addressing Allegations of Research Misconduct
RumeliDE adheres to COPE's Ethical Toolkit for a Successful Editorial. RumeliDE editors will take measures to prevent the publication of articles in which plagiarism, citation manipulation, data falsification, data fabrication, and other research misconduct have occurred. In no case will RumeliDE editors knowingly allow such misconduct to occur. If RumeliDE editors are aware of any allegations of research misconduct related to an article published in their journal, they will follow COPE's guidelines regarding allegations.
Ethical Violation Notifications
Readers can send an e-mail to editor@rumelide.com if they notice a significant error or inaccuracy in an article published in RumeliDE or if they have any complaints about the editorial content (plagiarism, duplicate articles, etc.). We welcome applications as it will provide an opportunity for us to improve, and we will respond quickly and constructively.
Correction, Retraction, Expression of Concern
Editors may consider publishing a correction if minor errors are detected in the published article that do not affect the findings, interpretations and conclusions. Editors should consider retracting the manuscript if there are major errors/violations that invalidate the findings and conclusions. Editors should consider issuing a statement of concern if there is a possibility of research or publication misconduct by the authors; there is evidence that the findings are unreliable and that the authors' institutions have not investigated the incident; or the potential investigation appears unfair or inconclusive. COPE and ICJME guidelines regarding correction, retraction or expression of concern are taken into account.
Publication of Studies Based on Surveys and Interviews
RumeliDE journal adopts the "Code of Conduct and Best Practice Guidelines for Journal Editors" and "Code of Conduct for Journal Publishers" principles of the Committee on Publication Ethics (COPE) in order to provide ethical assurance in scientific periodical publishing. In this context, the following points should be complied with in the studies submitted to the journal:
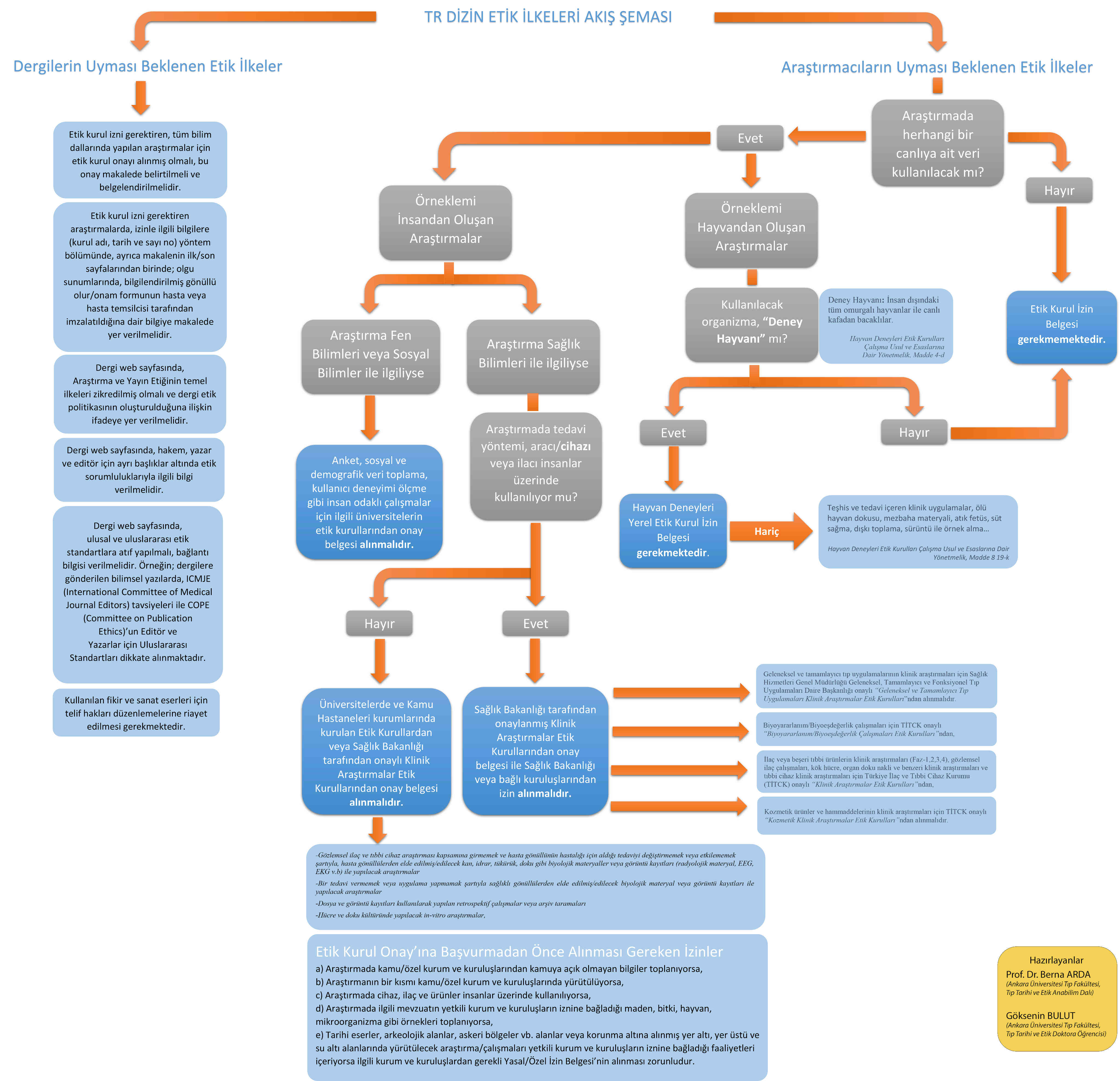
1) For research in all disciplines that require ethics committee approval (ethics committee approval must be obtained, this approval must be stated and documented in the article.
2) In studies requiring ethics committee authorisation, information about the authorisation (name of the committee, date and number) should be included in the method section and also on one of the first/last pages of the article; in case reports, information on the signature of the informed consent form should be included in the article.
Special Issue Publication Policy
Special issues can be published twice a year upon the request of the Editorial Board. Articles submitted for inclusion in the special issue are first subjected to a preliminary editorial review. Then, they are examined for compliance with the journal's spelling rules and similarity screening is performed to prevent plagiarism. After these stages, they are included in the peer review process using the double-blinding model.
Editorial Confidentiality Obligation
RumeliDE's editors treat all submitted articles as confidential documents, which means that they will not disclose information about an article to anyone without the authors' permission. During the article review process, the following people have access to the articles: Editors, Referees, Editorial Board Members. The only situation where details about a manuscript may be passed to a third party without the authors' permission is if the editor suspects serious research misconduct.
Allegations-Suspicions of Scientific Misconduct
There are different definitions of scientific misconduct. We address these issues on a case-by-case basis as RumeliDE follows the guidance established by the major publication ethics bodies. If the editor suspects or alleges an ethical violation, they are obliged to take action. This duty extends to both published and unpublished articles. The editor should not simply reject articles raising concerns about possible misconduct. He/she is ethically obliged to follow up on alleged cases. The editor should follow COPE flowcharts when appropriate. Editors should first seek a response from those suspected of misconduct. If they are not satisfied with the response, they should ask the relevant employer or organisation to investigate. The editor should make all reasonable endeavours to ensure that an appropriate investigation into the alleged misconduct is carried out; if this fails, the editor should make all reasonable attempts to insist on a resolution to the problem. This is an onerous but important task.
RumeliDE follows COPE's Ethical Toolkit for a Successful Editorial Office. RumeliDE editors will take measures to prevent the publication of articles in which plagiarism, citation manipulation, data falsification, data fabrication, and other research misconduct have occurred. In no case will RumeliDE or its editors knowingly allow such misconduct to occur. In the event that RumeliDE editors become aware of any allegations of research misconduct related to an article published in their journal, they will follow COPE's guidelines regarding allegations.
Reviewers should inform the Editor if they suspect research or publication misconduct. The Editor is responsible for taking the necessary action in accordance with COPE recommendations.
RumeliDE is committed to following the COPE flowcharts when faced with allegations of misconduct on the following or similar issues.
- What to do when re-publication is suspected
- What to do when plagiarism is suspected
- What to do if fabricated data is suspected
- Requests for change of authorship
- What to do if an undisclosed conflict of interest is suspected
- What to do if unfair or gift authorship is suspected
- What to do when an ethical problem is suspected in an article
- What to do when the suspicion of ethical violation is directly notified by e-mail, etc.
- What to do when a suspected ethical violation is announced via social media
Complaint Procedure
This procedure applies to complaints about content, procedures or policies that are the responsibility of RumeliDE or our editorial staff. Complaints can provide an opportunity and incentive for improvement and we aim to respond quickly, courteously and constructively.
The complaint must relate to content, procedures or policies that are the responsibility of RumeliDE or our editorial team. Complaints should be emailed directly to editor@rumelide.com and will be treated confidentially. The editor responds to complaints immediately. The editor follows the procedure outlined in the COPE flowchart regarding complaints.
Complaints are reviewed by the relevant member of the editorial team and if they cannot be resolved, the following processes are followed:
- If this initial response is considered inadequate, the complainant may request that the complaint be forwarded to a more senior member of the journal.
- If the complainant is not satisfied, the complaint may be forwarded to the editor-in-chief.
- If possible, a full response will be given within two weeks.
COPE publishes a code of practice for editors of scientific journals. This should facilitate the resolution of disputes with editors, journals and publishers, but only after the journal's own complaints procedures have been exhausted.
Appeal Process
We welcome serious objections to the evaluations made by editors and reviewers. If you feel that we have rejected your paper because we misunderstood its scientific content, please send an appeal message to our editorial team at editor@rumelide.com. Do not attempt to submit a revised version of your manuscript at this stage. If, after reading your appeal letter, we realise that your appeal is justified, we may invite you to submit a revised version of your manuscript. Your manuscript will then be resubmitted to the external reviewer process. Please include as much detail as possible in the appeal letter. Finally, we can only consider one appeal per manuscript, so please take the time and effort to write the letter in detail to make your appeal clear - you have one chance, so use it well. We have found that prolonged deliberation over rejected papers is often unsatisfactory for both authors and editors, so we do not process multiple appeals for the same paper.
Conflicts of Interest
A conflict of interest arises when professional judgement about a primary interest may be influenced by a secondary interest (such as financial gain or personal competition). We believe that we need to know the authors' competing interests in order to make the best judgement about how to handle an article, and that readers should know them if we publish the article.
Any interest, financial or otherwise, that may cause the person to be conflicted in his or her work, significantly impair his or her objectivity, or give an unfair advantage in favour of any person or organisation. All sources of financial support received during the conduct of the research and preparation of the manuscript and the role of sponsors in the study should be disclosed. If there is no source of funding, this should also be indicated. Examples of potential conflicts of interest that should be disclosed include consultancies, salaries, grants. Potential conflicts of interest should be disclosed at the earliest possible stage.
RumeliDE has a set process for handling submissions from editors, staff or editorial board members to ensure impartial review. Such submissions are first referred to other journals. If this is not possible, the owner of the submission will be suspended from the journal. These submissions are reviewed in a double blind process.
The editor should not be involved in decisions about manuscripts written by him/herself or family members. Furthermore, such a paper should be subject to all the usual procedures of the journal. The editor should follow the ICMJE guidelines on disclosure of potential conflicts of interest by authors and reviewers.
Principles of Research Ethics
RumeliDE observes the highest standards in research ethics and adopts the international research ethics principles defined below. It is the responsibility of the authors to ensure that manuscripts comply with ethical rules.
- The principles of integrity, quality and transparency must be ensured in the design, review of the design and conduct of the research.
- The research team and participants should be fully informed about the purpose of the research, its methods and possible uses; the requirements and risks, if any, of participation in the research.
- Confidentiality of information provided by research participants and confidentiality of respondents must be ensured. The research should be designed to protect the autonomy and dignity of the participants.
- Research participants should take part in the research voluntarily and should not be under any coercion.
- Harm to the participants should be avoided. The research should be planned in a way that does not put the participants at risk.
- Research independence must be clear and explicit; if there is a conflict of interest, it must be stated.
- In experimental studies with human subjects, the written informed consent of the participants who decide to participate in the research must be obtained. The consent of the legal guardian of children and those under guardianship or those with a recognised mental illness must be obtained.
- If the study will be carried out in any institution or organisation, approval must be obtained from this institution or organisation that the study will be conducted.
- In studies with a human element, it should be stated in the "method" section that "informed consent" has been obtained from the participants and ethics committee approval has been obtained from the institution where the study is conducted.
The legal responsibilities arising from the content of the studies published in the journal belong entirely to the author(s).
This journal
WMA Helsinki Declaration ethical principles (https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/);
ISMJE recommendations (https://www.icmje.org/recommendations/); (https://www.icmje.org/journals-following-the-icmje-recommendations/)
COPE (Committee on Publication Ethics/ https://publicationethics.org/guidance/Guidelines) has adopted the ethical rules.
Research requiring Ethics Committee approval: 1. All kinds of research conducted with qualitative or quantitative approaches that require data collection from participants using survey, interview, focus group study, observation, experiment, interview techniques, 2. Use of humans and animals (including materials/data) for experimental or other scientific purposes, 3. Clinical research on humans, 4. Research on animals.
Subjects requiring declaration: Retrospective studies in accordance with the law on the protection of personal data, 2. "Informed consent form" in case presentations, 3. Obtaining permission from the owners for the use of scales, questionnaires, photographs belonging to others, 4. Copyrights for intellectual and artistic works used.
This content was issued on 07.12.2023 and has been viewed for 996 times.


